
|
|
back |
|
Private
|

Electrolyte
Electrolyte
- ionic liquid or molten salt (may be mixed also with a solvent) [ii],[iii],[iv],[v],
- salts in aqueous solvents,
- salts in organic
solvents
The ionic liquid electrolytes are much more viscous than the liquid electrolytes made with a solvent. The ionic conductivity of ionic liquid is strongly reduced by the limited mobility of the ions with the consequence of a series resistance which is at least ten times bigger. The capacitance is also reduced, typical to half of that of an organic solvent. The reasons are on one hand that the ionic liquid ions are generally bigger and on the other hand, that the full carbon area is not available due to the very long migration time requested by the ions. The use of ionic liquids may be interesting at very high temperature where the series resistance is acceptable and where the other electrolytes would be beyond the solvent boiling temperature. Even if the ionic liquid decomposition voltage is expected to be equal to about 2.5 V at anodic potential and -4 V at cathodic potential, it tends to decompose and polymerize after several hundred charge/discharge cycles at cell voltage around 2 V. To bring ionic liquids into commercial use it's still necessary to investigate the window potential width at higher temperature where the activation energy is smaller and the behavior of the other material of the system in this extended potential window. The main brakes for the ionic liquids introduction in the industrial supercapacitors are their price and their conditioning.
The choice between aqueous and organic electrolyte [vi] depends on 4 parameters which are contradictory, the resistance, the capacitance, the manufacturability and the potential window size in which the system is electrochemically stable.
For given current frequency and intensity measurement conditions, the carbon capacitance density in the water is about twice that in an organic solvent while the series resistance is at least 10 times smaller.
The aqueous electrolytes have the advantage to be manipulated in atmospheric condition. The production process is simplified in the sense that the capacitive elements don't require any drying before the electrolyte impregnation and no protection against moisture contamination (glove box or dry room) must be undertaken during the impregnation. The main difficulties lay in the tightness capability of the housing closing system versus high acidic or basic liquids and in the cost of the current collector. The cheap aluminum doesn't sustain these conditions.
These electrolytes are unfortunately strictly limited by the maximum operating value of 1.23 VDC, which corresponds to the decomposition potential of the water molecules in hydrogen and oxygen gas. The organic electrolytes may be operated today up to voltages of 3 VDC at room temperature during thousands of hours, as it is the case for the acetonitrile based BCAP0350 supercapacitor [vii]. To take advantage of the organic electrolyte large potential window it's necessary to preserve all the products of the system from the presence of water. This is a serious drawback of these types of electrolyte.
The final choice is mainly driven by the solvent window potential size which leads for the organic electrolyte to a typical improvement of the energy and power density by a factor 5.
Within the class of organic electrolyte the
discussion is focused today on the use of acetonitrile
 (AN, http://en.wikipedia.org/wiki/Acetonitrile). This solvent provides 10 times
more ionic conductance than propylene carbonate (PC) in the low temperature
range of the specification [viii]. In 2004 AN has been declassified from toxic to harmful. Contrarily
to the rumor propagated by the Japanese supercapacitor
manufacturers, there is no interdiction of use in Japan for AN.
(AN, http://en.wikipedia.org/wiki/Acetonitrile). This solvent provides 10 times
more ionic conductance than propylene carbonate (PC) in the low temperature
range of the specification [viii]. In 2004 AN has been declassified from toxic to harmful. Contrarily
to the rumor propagated by the Japanese supercapacitor
manufacturers, there is no interdiction of use in Japan for AN.
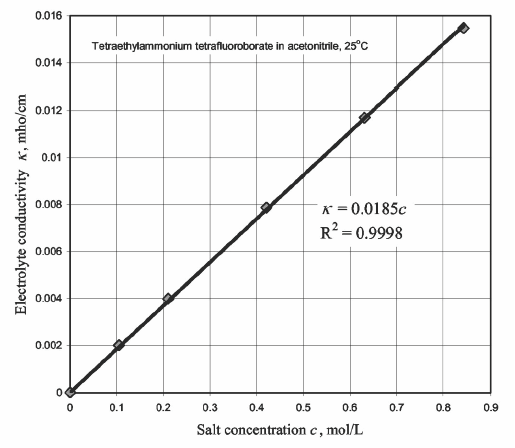
The ionic conductivity is provided by the addition of a salt in the solvent.
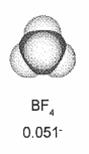
The most used salt is the tetraethylammonium tetrafluoroborate
(C2H5)4NBF4
because of its small size.
4N.jpg) Each molecule disassociates in a positive cation [(C2H5)4N]+
and in a negative anion [BF4]-. The electrolyte requirement
Each molecule disassociates in a positive cation [(C2H5)4N]+
and in a negative anion [BF4]-. The electrolyte requirement
 is to provide the highest ionic conductivity in the full specified temperature
range. In this domain, it must neither freeze nor vaporize, the salt must not
precipitate and the potential window must remain large enough to allow deep
cycling. The salt concentration must be high enough to avoid the ion depletion
at high voltage [ix]. A typical concentration for (C2H5
)4NBF4 in AN is 1 molar.
is to provide the highest ionic conductivity in the full specified temperature
range. In this domain, it must neither freeze nor vaporize, the salt must not
precipitate and the potential window must remain large enough to allow deep
cycling. The salt concentration must be high enough to avoid the ion depletion
at high voltage [ix]. A typical concentration for (C2H5
)4NBF4 in AN is 1 molar.
Electrolyte performance
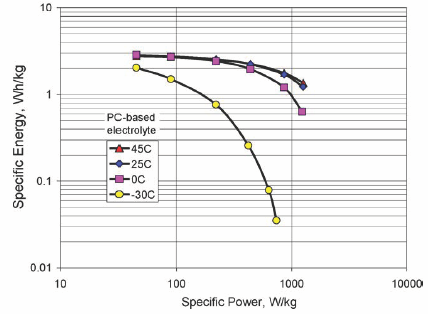
It has been shown [viii] that above 25C the supercapacitor capacitance is 5 to 10% bigger with AN in comparison to that obtained with PC. In the same temperature conditions the series resistance with PC is 50% bigger. At lower temperature the situation is even worse. At -30C for example, the capacitance is 50% bigger with AN and the series resistance is 5 times bigger with PC.
Source: Garmanage: Roland Gallay
[i]Ue M, Ida K, Mori S. Electrochemical properties of organic liquid electrolytes based on quaternary onium salts for electrical double-layer capacitors. J Electrochem Soc 1994;141 (11):2989-95.
[ii] McEwen AB, Ngo HL, Lecompte K, Goldman JL. Electrochemical properties of imidazolium salt electrolytes for electrochemical capacitor applications, J Electrochem Soc 1999;146 (5):1687-95.
[iii] Ue Makoto. Application of ionic liquids to double-layer capacitors, in Electrochemical aspects of ionic liquids. John Wiley & Sons; 2005, ISBN 9780471648512.
[iv] Balducci A, Soavi F, Mastragostino M. The use of ionic liquids as solvent-free green electrolytes for hybrid supercapacitors. Applied Physics A 2006;82:627-32.
[v] Kurzweil P, Chwistek M, Gallay R. Capacitance Determination and Abusive Aging Studies of Supercapacitors Based on Acetonitrile and Ionic Liquids. Proc. 16th International Seminar On Double Layer Capacitors, 2006, Deerfield Beach, USA, p. 78.
[vi] Tanahashi I, Yoshida A, Nishino A. Comparison of the electrochemical properties of electric double-layer capacitors with an aqueous electrolyte and with a nonaqueous electrolyte. Bull Chem Soc Jpn 1990;63:3611-4.
[vii] BCAP0350 supercapacitor from Maxwell Technologies. 350F, 3.2 mOhm, http://www.maxwell.com, 2004.
[viii] Liu P, Soukiazian S, Verbrugge M. Influence of temperature and electrolyte on the performance of activated-carbon supercapacitors. J Power Sources 2006;156:712-8.
[ix] Zheng JP, TR Jow. The effect of salt concentration in electrolytes on the maximum energy storage for double layer capacitors. J Electrochem Soc 1997;144 (7):2417-20.