
|
|
back
MENU
|
|
Private
|

Equations
Simulation: electrode model
Variables & Constantes
- ρ(x,t) electrical charge density [C/m3]
- J(x,t) current density= charge flux [A/m2]
- n(x,t) concentration particles [1/m3]
- j(x,t) particlesflux [1/m2s]
- c(x,t) molar concentration part. [mol/m3]
- N(x,t) molar flux [mol/m2s]
- Avogadro constant A = 6 1023 [1/mol]
- Electronic charge e = 1.6 10-19 [C]
- Faraday constant F = 96’500 [C/mole]
Charges
There are 4 charge species to consider a priori in an electrochemical device:
a) Positive and Negative ions in the electrolyte in the pores and in the separator.
b) Electrons and holes in the electrode (solid electronic conductors).
Electrode sketch
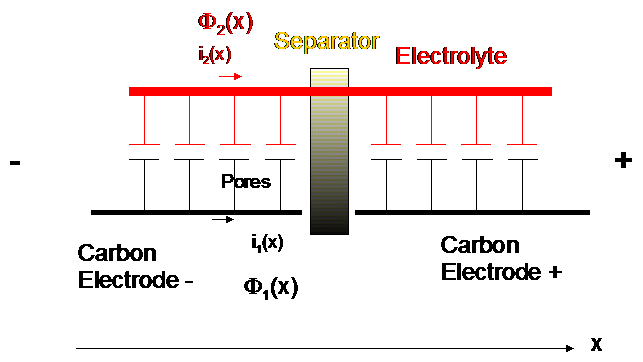
Resistance is much smaller in carbon => the carrier path will be maximized in it.
At high frequency only the pores close to the electrode surface will be reach
| Current definition: | 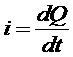 |
| Continuity equation: | 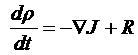 |
| Charge density: | 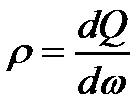 |
| Current density: | 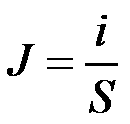 |
Electrode model
In each electrode volume element there is a mixing of two homogeneous phases: the electrode matrix and the pores.
Φ1(x,t) is the electric potential in the matrix.
Φ2(x,t) is the electric potential in the electrolyte “far enough” from the pore surface. We aren’t studying the potential in the double layer….
Φ1(x,t) and Φ2(x,t) depend on the position in the depth of the electrode.
i(x) = i1(x) + i2(x) where i1 is the current in the electrode matrix and i2 is the current in the electrolyte
Current in the electrode matrix
Only electronic conduction in the electrode matrix (carbon),
No diffusion.
Ohm law => i1: ![]()
Gradient definition => Φ
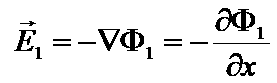
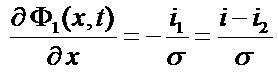
![]()
Liquid phase physical laws
Two laws:
Electrical field Coulombic force =>
Ohm law
Charge migration 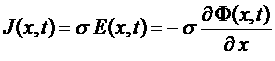
Concentration gradient => Fick law
Material diffusion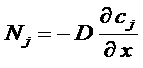
(multiplied by the charge) to get charges diffusion
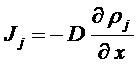
Opposed to the migration
Diffusion
Einstein relation 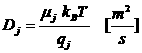
μ mobility [m2/Vs]
Brownian μp = vd/ F. Electrical μe = vd/ E.kB Boltzmann’s constant
T temperature
-
Gas R = A kB
Density n = z A c
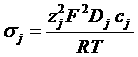
Current density in the liquid phase
Current density of species j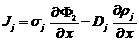
With Einstein relation
concentration flow
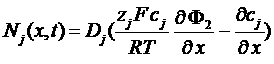
charges flow
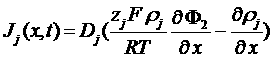
Neutrality of the solution
Far from the double layer the concentration of positive and negative ions in the electrolyte is equal
Drude model ![]()
σ conductivity
n density of carriers
zj Nr of charge of specie j
c charge molar concentration
Faraday F = A e
c+ = c- = c
ρ = 0
Single charge carrier
In most of the case the electrolyte salt is made of single charge ions
z+ = -z- = 1
J+ = F N+
J- = -F N-
i2 = J+ + J- = F (N+ - N-)
Total current in the electrolyte
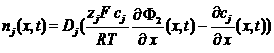
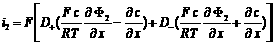
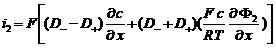
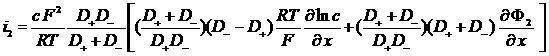
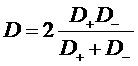
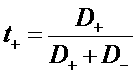
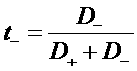
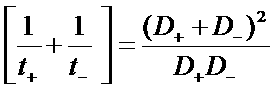
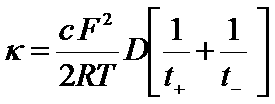
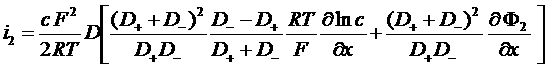
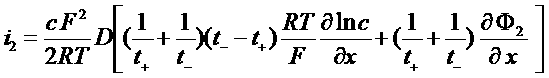
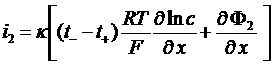
![]()
Charge storage at the interface
At the interface between the pores and the electrolyte there is a uniform capacitance C = C(x) per unit of pore surface.
The interface surface per unit of volume is given by a
The current involved per unit of volume is J(x,t)
The current of specie j Jj(x,t)
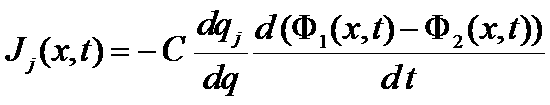
Interface neutrality
dq change of interface electrode charge
dq+ change of positive ion charge
dq+ /dq = dq- /dq = -1/2
Bring 4 electrons on the electrode surface => bring 2 negative ions and remove 2 positive ions in the electrolyte neighborhood.
Current of charge
J = (J+ + J-)
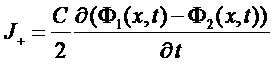
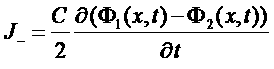
=> 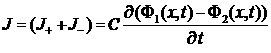
C capacity per pore surface unit
Continuity equation
Consider an electrode volume unit,
Its capacitance is given by aC, where a is the pore surface per electrode volume unit and C is the capacitance per surface unit
The current which flow through the unit surface (cross section of the unit volume) is i2
div i2 = [i2(x+δx)-i2(x)] / δx
dρ/dt = charge trapped in the double layer = aC dΦ2/dt
Continuity equation at the interface
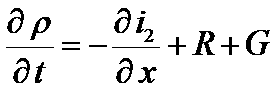
=> 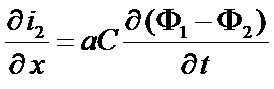
and => 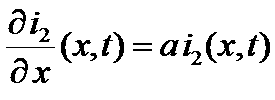
Equations in the electrode
Electronic conduction : 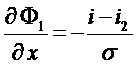
Ionic conduction: 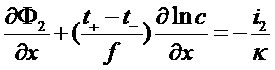
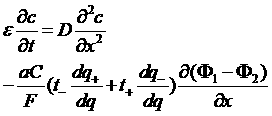
Equations in the separator
No electronic conduction (except selfdischarge through the separator)
i1(x,t) = 0
![]()
Field equations + Boundaries
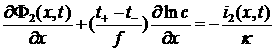

Continuity equations
G : charge separation (generation)
R : charge recombination
![]()
![]()